sparky
Well-known member
I've been doing a lot of work on anodic protection systems for my job, and I figured that I'd share the info with my fellow CP'ers.
There is a lot of misinformation going around about protecting your engine/hull/bracket/tabs, etc from galvanic corrosion. Most people understand that you have to protect underwater metals from stray current, but don't understand how to do so properly.
Basically, every metal has an electric potential. Some are higher (gold, bronze, brass, SS, etc), and some are lower (aluminum, zinc, magnesium). When you connect these differing metals in a conductive environment (seawater, fresh water, or brackish water), the more noble metal becomes the cathode, and the less noble metal becomes the anode in a very weak, primitive battery. Any of you ever make a clock than ran on a lemon with a zinc strip and a copper strip embedded in it? Same thing.
Here's a good chart of metal potentials:
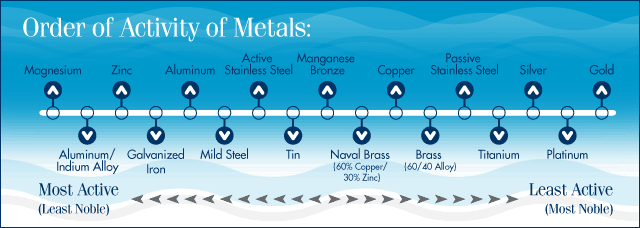
To understand what's going on with your situation:
1. Find the different metals on your boat. Prop, shafting, bracket, outdrive/outboard engine, trim tabs, exposed fasteners, and transducer.
2. Locate each metal on the chart. In our case (2520 w/ a F225), we've got Stainless (Prop, tabs, and exposed fasteners), bronze (transducer), and aluminum (bracket and outboard).
3. See which metal is the least noble (left-most) on the above chart. That's the metal that's going to turn into the anode without any protection. The other metals will steal electrons from that item, and it will corrode.
In our case, that'd be the motor and the bracket. We'd prefer that those don't corrode. The answer is to install something else to be the anode, something made of a metal that's less noble (lower electrical potential) than anything else on the boat. Many people think the answer is simple:
Zinc.
Well, zinc is less noble than mostly everything, but it has some major problems. First of all, if you take a zinc into fresh water, it'll generate an oxide layer, which effectively insulates it from the hull, and prevents it from doing anything (except looking nice). Secondly, if you haul your boat, and let the zinc dry out, it'll insulate itself in much the same fashion. Again, useless at protection.
Ok, how about magnesium. Magnesium is the least noble of the elements listed in the chart, so why not just use that? Well, it'll protect your metals for sure, but it can overprotect aluminum. When people talk about overprotection, they are referring to the generation of hydrogen bubbles on the cathode, similar to what happens in Hydrolysis. Basically, the electrical potential is strong enough that the water molecules split, generating hydrogen at the cathode and oxygen at the anode. This hydrogen is generated on a molecular scale, and will lift the paint on your aluminum outdrive or bracket. Plus, magnesium will very rapidly dissolve in brackish or sea water.
This leaves aluminum, but not just any aluminum. There are special alloys which include zinc and trace amounts of a rare metal called indium. The indium keeps the aluminum from making an oxide layer when wet or dry, and the zinc helps lower its potential to below that of structural (aircraft) or marine aluminum alloys.
Here's a chart of acceptable metals/hulls/motors/water types:
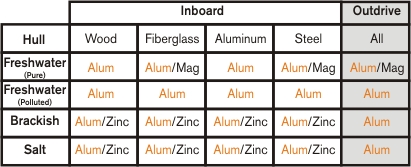
In any case, pretty much everyone here should be running aluminum anodes on their engines, hulls, brackets, and trim tabs. If you mix and match anode materials, you'll run into problems. Mercury is aware of this issue, and won't cover corrosion damage on outdrives or outboards if you use zinc anodes. Yamaha factory anodes are aluminum, and should always be replaced with aluminum.
Feel free to ask more questions regarding corrosion.
There is a lot of misinformation going around about protecting your engine/hull/bracket/tabs, etc from galvanic corrosion. Most people understand that you have to protect underwater metals from stray current, but don't understand how to do so properly.
Basically, every metal has an electric potential. Some are higher (gold, bronze, brass, SS, etc), and some are lower (aluminum, zinc, magnesium). When you connect these differing metals in a conductive environment (seawater, fresh water, or brackish water), the more noble metal becomes the cathode, and the less noble metal becomes the anode in a very weak, primitive battery. Any of you ever make a clock than ran on a lemon with a zinc strip and a copper strip embedded in it? Same thing.
Here's a good chart of metal potentials:

To understand what's going on with your situation:
1. Find the different metals on your boat. Prop, shafting, bracket, outdrive/outboard engine, trim tabs, exposed fasteners, and transducer.
2. Locate each metal on the chart. In our case (2520 w/ a F225), we've got Stainless (Prop, tabs, and exposed fasteners), bronze (transducer), and aluminum (bracket and outboard).
3. See which metal is the least noble (left-most) on the above chart. That's the metal that's going to turn into the anode without any protection. The other metals will steal electrons from that item, and it will corrode.
In our case, that'd be the motor and the bracket. We'd prefer that those don't corrode. The answer is to install something else to be the anode, something made of a metal that's less noble (lower electrical potential) than anything else on the boat. Many people think the answer is simple:
Zinc.
Well, zinc is less noble than mostly everything, but it has some major problems. First of all, if you take a zinc into fresh water, it'll generate an oxide layer, which effectively insulates it from the hull, and prevents it from doing anything (except looking nice). Secondly, if you haul your boat, and let the zinc dry out, it'll insulate itself in much the same fashion. Again, useless at protection.
Ok, how about magnesium. Magnesium is the least noble of the elements listed in the chart, so why not just use that? Well, it'll protect your metals for sure, but it can overprotect aluminum. When people talk about overprotection, they are referring to the generation of hydrogen bubbles on the cathode, similar to what happens in Hydrolysis. Basically, the electrical potential is strong enough that the water molecules split, generating hydrogen at the cathode and oxygen at the anode. This hydrogen is generated on a molecular scale, and will lift the paint on your aluminum outdrive or bracket. Plus, magnesium will very rapidly dissolve in brackish or sea water.
This leaves aluminum, but not just any aluminum. There are special alloys which include zinc and trace amounts of a rare metal called indium. The indium keeps the aluminum from making an oxide layer when wet or dry, and the zinc helps lower its potential to below that of structural (aircraft) or marine aluminum alloys.
Here's a chart of acceptable metals/hulls/motors/water types:

In any case, pretty much everyone here should be running aluminum anodes on their engines, hulls, brackets, and trim tabs. If you mix and match anode materials, you'll run into problems. Mercury is aware of this issue, and won't cover corrosion damage on outdrives or outboards if you use zinc anodes. Yamaha factory anodes are aluminum, and should always be replaced with aluminum.
Feel free to ask more questions regarding corrosion.





